Via Electronic Transmission:
http://www.regulations.gov
rcra-docket@epa.gov
TO:
RCRA Docket Officer
Environmental Protection Agency Mail Code: 2822T
1200 Pennsylvania Avenue, N.W.
Washington, D.C. 20460
FROM:
Charlotte A. Smith, R. Ph., M.S.
Senior Regulatory Advisor
PharmEcology Services
P.O. Box 1654
Brookfield, WI 53008-1654
September 1, 2023
RE: EPA Docket No. EPA-HQ-OLEM-2023-0081. Hazardous Waste Generator Improvements Rule, the Hazardous Waste Pharmaceuticals Rule, and the Definition of Solid Waste Rule; Technical Corrections
Dear Docket Officer:
PharmEcology Services, a business unit of WM Sustainability Services, has been involved with the regulation of hazardous waste pharmaceuticals since its inception as a private company in 2000 as PharmEcology, LLC. We therefore read with great interest the changes proposed in the Technical Corrections published in the Federal Register, Vol.88, No. 152, pages 54086 through 54115 and have the following comments.
- 1. In Table 1, page 54092, EPA lists "Examples of Hazardous Waste Pharmaceuticals Listed in Appendix IV to Part 268 – Wastes Excluded from Lab Packs Under the Alternative Treatment Standards of 268.42(c)." These include the following which we do not believe should be listed as they are not relevant to healthcare:
D009 Mercury (Toxicity characteristic): Mercury as a preservative always occurs as part of an organic molecule: thimerosal (C9H9HgNaO2S), phenylmercuric acetate (CH3COOHgC6H5) (28.53% carbon), and phenyl mercuric borate (C6H7BHgO3). Calomel (Hg2Cl2) is no longer in active use and always occurs as a powder and would not be used as such in a finished dosage form pharmaceutical.
P076 Nitric oxide: Is this perhaps a reference to Nitrous Oxide, N2O, also known as "laughing gas," which is not listed in Appendix IV? Nitric oxide has no known medical use to our knowledge and thus is confusing.
U151 Mercury: Mercury is no longer used as a sole active ingredient in any medication. The salts of mercury, bichloride and iodide, are only available as bulk chemicals and if used at all would be imbedded in some type of organic base.
We agree that P012 – Arsenic trioxide is a current pharmaceutical entity and is an appropriate example.
- In the section "Marking Lab Packs for Shipping," page 54092 column 1, the statement is made: "However, be aware that the shipping standards for non-creditable and evaluated hazardous waste pharmaceuticals require that lab packs containing D004 (arsenic), D005
(barium), D006 (cadmium), D007 (chromium), D009 (lead), D010 (selenium) or D011 (silver) must be marked with the EPA hazardous waste numbers." (Bolding added.) Is this requirement intended only for reverse distributors since non-creditable and evaluated hazardous waste pharmaceuticals are used in the same sentence or does this also apply to healthcare "lab packs" being shipped through hazardous waste haulers for disposal at a RCRA permitted incinerator? If this does apply to healthcare lab packs, we believe this is a new interpretation of the regulations and needs to be emphasized and added to the FAQs. This should also be included in any revisions of the 10-Step Blueprint for Managing Pharmaceutical Waste in US Healthcare Facilities.
Also, due to its toxicity, lead, D009, is never used as a pharmaceutical and would only be generated from the radiology department in the form of lead aprons or from other sources within the facility and would not fall under the Pharmaceutical Rule. The only drug
containing arsenic is arsenic trioxide which is P-listed so it would not be listed twice. If this does apply to healthcare "lab packs" being shipped to hazardous waste haulers, this is new information. Regardless, we believe it's important to avoid confusion by not listing chemicals that would not be present in pharmaceuticals.
- On page 54109 column 1, 262.11 (g)…the generator also must mark its containers with all applicable EPA hazardous waste numbers (EPA hazardous waste codes) according to 262.32.
Consider a reference to healthcare facilities and Subpart P and include a reference to the exclusion for hazardous waste pharmaceuticals to avoid confusion.
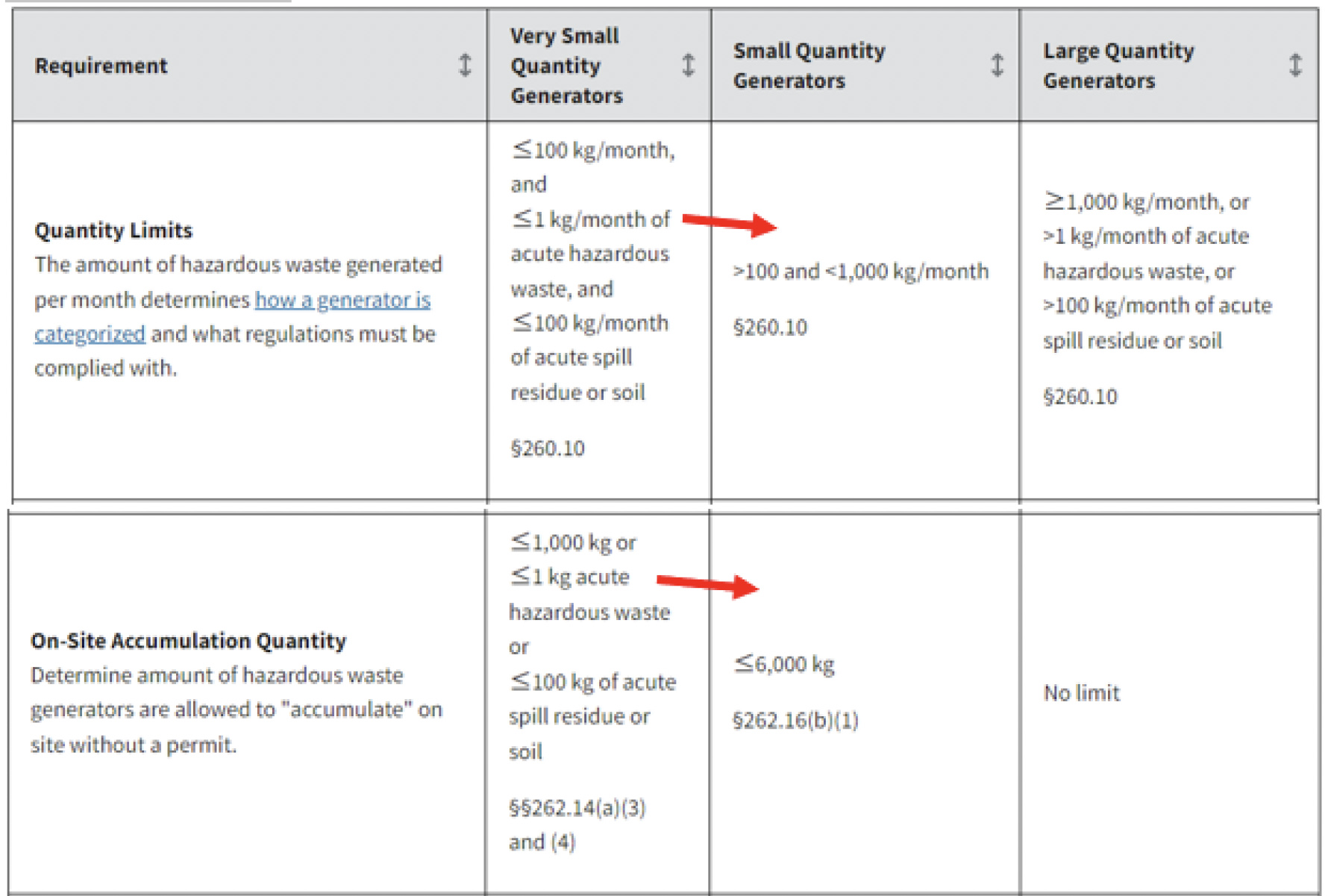
- On page 54109, column 3: 262.13 Conditions for exemption for a small quantity generator that accumulates hazardous waste. …(b) (1) Accumulation limit. The quantity of acute hazardous waste accumulated on site never exceeds 1 kg (2.2 pounds) and the
quantity of non-acute hazardous waste accumulated on site never exceeds 6,000 kg (13,200 pounds).
Consider updating the SQG Quantity Limits per month and the SQG On-Site Accumulation Quantity fields of the Generator Summary Table to specify less than or equal 1 kg acute hazardous waste for SQGs.
https://www.epa.gov/hwgenerators/hazardous-waste-generator-regulatory-summary
- While not officially part of this regulatory action, we have noticed that the drug "phenobarbital" continues to be used as an example of hazardous waste pharmaceuticals that are also controlled substances. After researching current dosage forms of
phenobarbital, which itself is not ignitable, all the elixirs on the market contain less than 24% alcohol1,2 which meet the following exemption as an ignitable hazardous waste: 40 CFR § 261.21 Characteristic of ignitability.
(a) A solid waste exhibits the characteristic of ignitability if a representative sample of the waste has any of the following properties:
(1) It is a liquid, other than a solution containing less than 24 percent alcohol by volume and at least 50 percent water by weight, that has a flash point less than 60 °C (140 °F), as determined by using one of the following ASTM standards: ASTM D93–79, D93–80,
D3278–78, D8174–18, or D8175–18 as specified in SW–846 Test Methods 1010B or 1020C (all incorporated by reference, see § 260.11 of this subchapter). We therefore request that a clarification be placed in the final regulation and in the pertinent FAQs to correct this reference to avoid further confusion in the disposal of all dosage forms of phenobarbital.1https://archive.hshsl.umaryland.edu/bitstream/handle/10713/7776/PPAG%20Alcohol%20Content%20in%20Peds%20Meds%20
Poster-4-10-18.pdf?sequence=1&isAllowed=y
2 https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=29be31ef-108e-4b45-9ecc-b6d671da2826



